 Education & Careers
Education & Careers
Can't find what you're looking for? Contact us
For better functionality and user experience, Invibio.com uses cookies which can be disabled at any point by adjusting your browsers settings. By using this website you consent to our use of these cookies. Please view our Cookie Policy for more information
 Education & Careers
Education & Careers
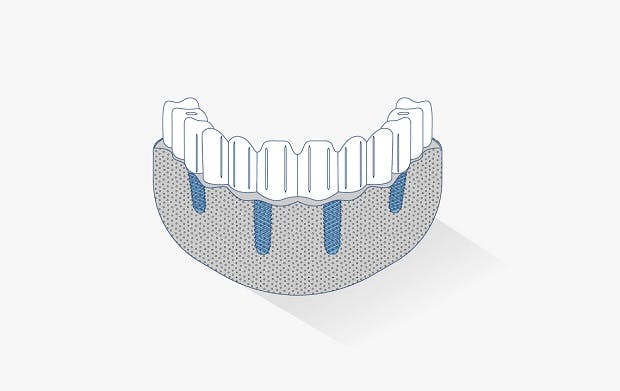
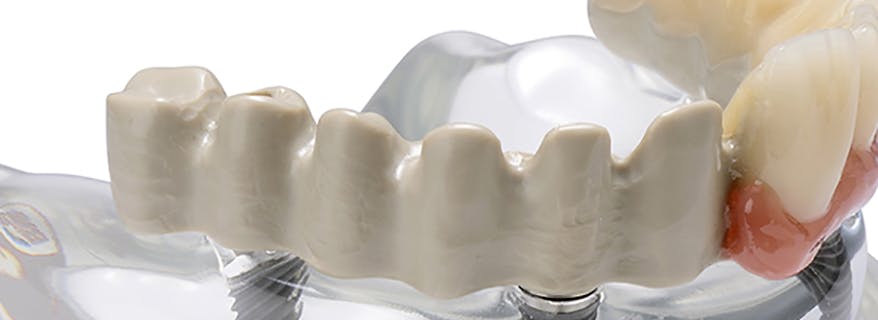

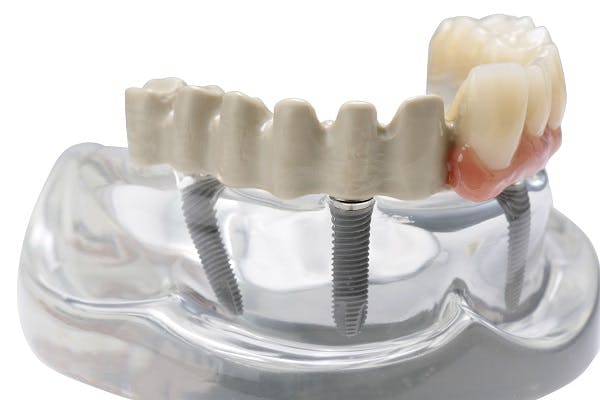
The JUVORA Dental Disc is suitable for a wide range of indications, including removable clasp dentures, removable attachment dentures and telescope dentures. Read the eIFU for guidance on intended use.

Visit our resource centre for instructions on how to use the JUVORATM Dental Disc


References
1. de Araújo Nobre, M.; Moura Guedes, C.; Almeida, R.; Silva, A.; Sereno, N. Hybrid Polyetheretherketone (PEEK)–Acrylic Resin Prostheses and the All-on-4 Concept: A Full-Arch Implant-Supported Fixed Solution with 3 Years of Follow-Up. J. Clin. Med. 2020, 9, 2187.
2. “In-mouth comfort” comparison: 6 - Malo Clinic, Miguel de Araujo Nobre et al. Journal of Clinical Medicine 2020
3. “Quality of life” comparison – OHRQoL and OHIP-20 score: 4 - Sheffield University, Zaid Ali et al, Pilot study comparing PEEK to COCr as RPDs frameworks, International Journal of Prosthodontics, 2020
4. “Quality of life” comparison – OHRQoL and OHIP-14 score: 3 - PEEK Biomaterials Handbook 2nd Ed 2019 chapter 20 Dental
5. ALL-ON-4 is a trademark of Nobel Biocare Services AG
From the invention of PEEK over 40 years ago, Victrex has continually pioneered new PAEK-based polymers, materials and solutions that have transformed markets, delivering global impact in the toughest environments.
We bring transformational & sustainable solutions that address world material challenges every day.